Our Nanoparticle Platform
Nanoparticle technology now comprises a very large field, even within the pharmaceutical and diagnostic space. Most in vivo nanoparticle developments have been focused on the delivery of drugs or diagnostic signals to specific tissues. In these instances, the vascular system is merely a delivery conduit, with the target tissues or cells positioned outside of vessels. Consequently, most formulations have utilized particle sizes small enough to escape through capillary junctions or allowed the release of active agent from the particle once in the vicinity of the target.
HMRI’s nanoparticle platform has several characteristics not usually found in these other formulations. Our composition is designed to remain within the vascular system, in effect targeting blood tissue. It is also designed to be immunologically inert, with minimal uptake by macrophages. Finally, its size distribution is not filterable by kidney glomerulae, preventing renal clearance, and, with its lack of reticulum endothelial system uptake, the particles are safely cleared via the hepatobiliary system. This makes the technology a close approximation to a one-compartment model of biodistribution, a rarity.
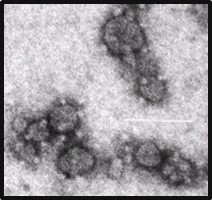
There are numerous healthcare applications for a technology such as this, but HMRI has chosen contrast MRI as the most compelling initial product application for the platform. For this application the Company has selected a novel particle comprised of: a phospholipid; a PEGylated phospholipid; and a lipid-conjugated metal chelate. The particles are approximately 50-70nm in size, and display chelated gadolinium on their surface (Figure 1).
Nanobrite™ is a disruptive innovation, with excellent patent protection, that is being developed to dramatically improve the value of MRI.

Nanobrite™ Features
– Superior Signal Enhancement -- Ultra high MRI definition of blood vessels with relaxivity of 134 vs 4 to 28 for other GBCAs
– Longer useful plasma time = viable Image time – 90 to 120 minutes versus 4 to 36 minutes with other GBCAs
– Non-renal elimination – provides a more safe alternative to patients with renal insufficiency. Nanobrite™ is cleared through the liver
– Lower doses requirement – due to the greater signal strength, lower doses of Nanobrite™ are required thus increasing the safety margins
Superior images at lower doses than commercially available GBCAs
